Scope of the project
New generation biocarriers
The continuous upgrading and implementation of new stricter water quality regulations as well as the discovery of new advanced wastewater treatment processes have made the mature technology of biofilm growth reactors on biocarriers in the aerobic and anaerobic treatment of municipal wastewater one of the current treatment trends of wastewater.
Biocarriers provide the possibility of immobilizing microorganisms on their surface, thus improving the efficiency of biological processes. These are aggregates, materials with different shapes and sizes and are usually made of porous ceramics, polyethylene, polypropylene, etc. The adhesion of microorganisms to the surface of the biocarrier and their subsequent growth creates a dense biofilm on the surface but also inside the carrier with a thickness of a few millimeters. The material that binds and binds the microorganisms are extracellular polymers produced by the microorganisms. The nature of the biocarrier material, specifically its surface texture and porosity, has a significant impact on the performance of the biofilm, as it significantly affects the rate of attachment and growth of microorganisms. Natural and readily available materials are often used as biocarriers, where they do not achieve satisfactory results in the performance of biological processes, due to their reduced specific surface area and therefore show an inability to contain many colonies of microorganisms, compared to commercial biocarriers.
The main objective of 3DBiofilm is the evolution of biocarriers from their simple form, i.e. as an inert material for the immobilization of microorganisms, to new generation biocarriers, where by using new materials for their construction through 3D printing (3DP), they will new properties will be given and the complexity of their shapes will increase so that they can facilitate heterogeneous biofilm development, through the creation of different paths of diffusion and transport of the dissolved components, and they will be able to stimulate the metabolism of the communities of microorganisms (colonists), achieving a higher efficiency of wastewater treatment and at the same time maintaining smoothness and stability in the functioning of biological processes.
3D printing (3DP) technology is a new technology used in the rapid prototyping (RP) industry. Also known as additive manufacturing, it is based on the successive layering of homogeneous materials or alloys to manufacture tool prototypes with absolute precision, speed and reduced manufacturing costs. Accuracy is ensured by previously designing a 3D digital model using software programs to calculate the dimensions and predict the mechanical and physical properties of the object to be printed.
The 3DP technique overcomes the limitations of classical industry, as it can combine different raw materials regardless of the deviations in their chemical and physical properties and enables rapid automated manufacturing. Therefore, through this technique, the desired size and shape of the biocarriers can be designed and determined with the aim of the largest possible specific surface, as well as the appropriate printing material can be selected by determining its physicochemical characteristics, achieving ideal biocarrier-microorganism affinity conditions.
Zeolite and aluminosilicate materials
Zeolites and aluminosilicate materials are abundant in Greece, are important low-cost materials and are particularly preferred for wastewater treatment. As far as anaerobic wastewater treatment is about, these materials potentially provide an excellent working environment for microorganisms involved in either the biomass hydrolysis process or the methane formation process. Recent studies have demonstrated the ability of zeolites to be colonized by certain microbial populations during anaerobic operation. In addition, it was shown that microorganisms acting on biomass as decomposers, obtained by selective cultivation, can be immobilized on zeolite surfaces. At the same time, zeolites show a remarkable improvement in treatment performance in aerobic wastewater treatment systems as well, due to their excellent ion exchange properties, their adsorption capacity and their ability to be used for sieving molecules/components.
Extrusion of zeolite monoliths has already been described in the literature. However, the effect of different binders on the extrusion behavior for 3DP zeolites and the effect on the mechanical and physicochemical properties is not fully understood. Some early investigations of 3D printing biocarriers from zeolite paste have already been carried out by Thakkar et al. (2016) who fabricated 3DP biocarriers from 13X and 5A zeolite monoliths. 3DP biocarriers from novel materials are a promising innovative technology, still in its early stages, that may provide increased benefits in a variety of applications beyond waste treatment optimization. The 3DBiofilm project will use a rapid prototyping 3D printing method that will allow testing different zeolite-linkage system compositions, as well as scanning different architectural monoliths.
Application of biocarriers for the optimization of anaerobic waste treatment
The type of reactor that finds application in large-scale biogas units is the Continuous Stirred Tank Reactors (CSTR). In CSTRs, continuous stirring is intended to thoroughly mix the fresh substrate with the active digested contents of the reactor. The hydraulic retention time (HRT) is kept sufficiently high to achieve satisfactory degradation of the most slowly degradable portion of the substrate, otherwise the loss of undegraded substrate must be accepted. This shortcoming of CSTR reactors can be addressed by the addition of biocarriers with the possibility of tethered suspension proposed by this research proposal. The formation of biome on the surface of the biocarriers will significantly accelerate the degradation rate of the new substrate significantly reducing the HRT. In addition, it will increase the effectiveness of CSTR reactors in blocking effects from toxic substances such as ammonia, etc., due to the achievement of a stable microbial community. In addition, in the Upflow Anaerobic Sludge Blanket UASB reactors, the use of the new biocarriers, in addition to the larger specific surface (compared to conventional ones) that will distinguish them resulting in the immobilization of a greater number colonies of microorganisms, their construction from new materials, will enhance the process of anaerobic digestion, due to the ability of the specific materials, to bind ammonium and/or other ions (based on modification), preventing the reduction of methane production from the frequent occurrence of the phenomenon of ammonia toxicity.
The simplest method of applying biocarriers for aerobic waste treatment is the MBBR Moving Bed Biofilm Reactors, which are also found in combination with filtration membranes (MBBR-MBR-Moving Bed Biofilm Reactors-Membrane Bioreactors). Modern MBBR or MBBR-MBR methods use freely moving submerged biocarriers in aerated tanks, combining two different processes, suspended and attached biomass growth. Biocarriers increase the efficiency of biological treatment by achieving high biodegradation and therefore better treatment efficiency of the waste, which can then be disposed of to the final recipient and/or reused. At the same time, they reduce the problem of clogging of the filtration membranes by scraping their surface. This technology does not require the continuous addition of expensive reagents or the production of hazardous residues, making it highly competitive from an economic and environmental point of view. Therefore, it is a highly advanced technology with low capital and operating costs, while at the same time it is simple, unified, reliable and stable in its operation.
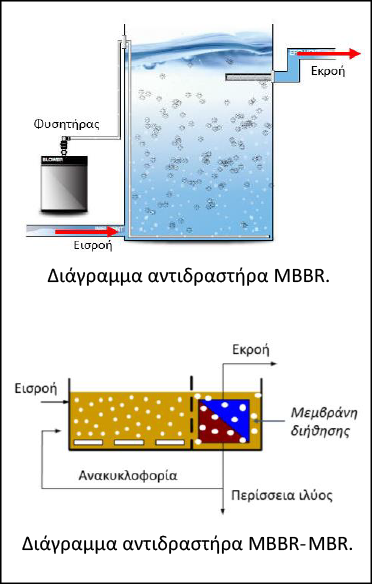
According to research, it has been identified that the most critical parameter determining the performance of MBBR and MBBR-MBR technology is the appropriate design of the biocarrier in terms of specific surface area, material, texture and orientation. The technology of 3DP is the one that can provide the solution to the issue of the construction of the optimal biocarrier, providing flexibility in the design and the possibility of choosing its construction material.
Objectives of 3DBiofilm
- Design and development of advanced biocarrier geometries per industrial application (aerobic-anaerobic)
- Preparation of modified materials using phyllomorphic aluminosilicates, zeolites mixed with starch, saccharides, etc. to create new innovative materials that will find application as 3DP printing pastes (International innovation)
- Printing of innovative 3D-biocarriers per industrial application (aerobic – anaerobic) with “augmented” features and properties (International innovation)
- Exploitation of low-cost minerals found in abundance in our country, to create a new market not only nationally but internationally
- Acquiring know-how in the newly-emerging three-dimensional printing (3DP) technology for the production of prototypes and advanced biocarriers in a Greek private enterprise and in a Greek human resource in collaboration with a cutting-edge research center in the field of 3DP

